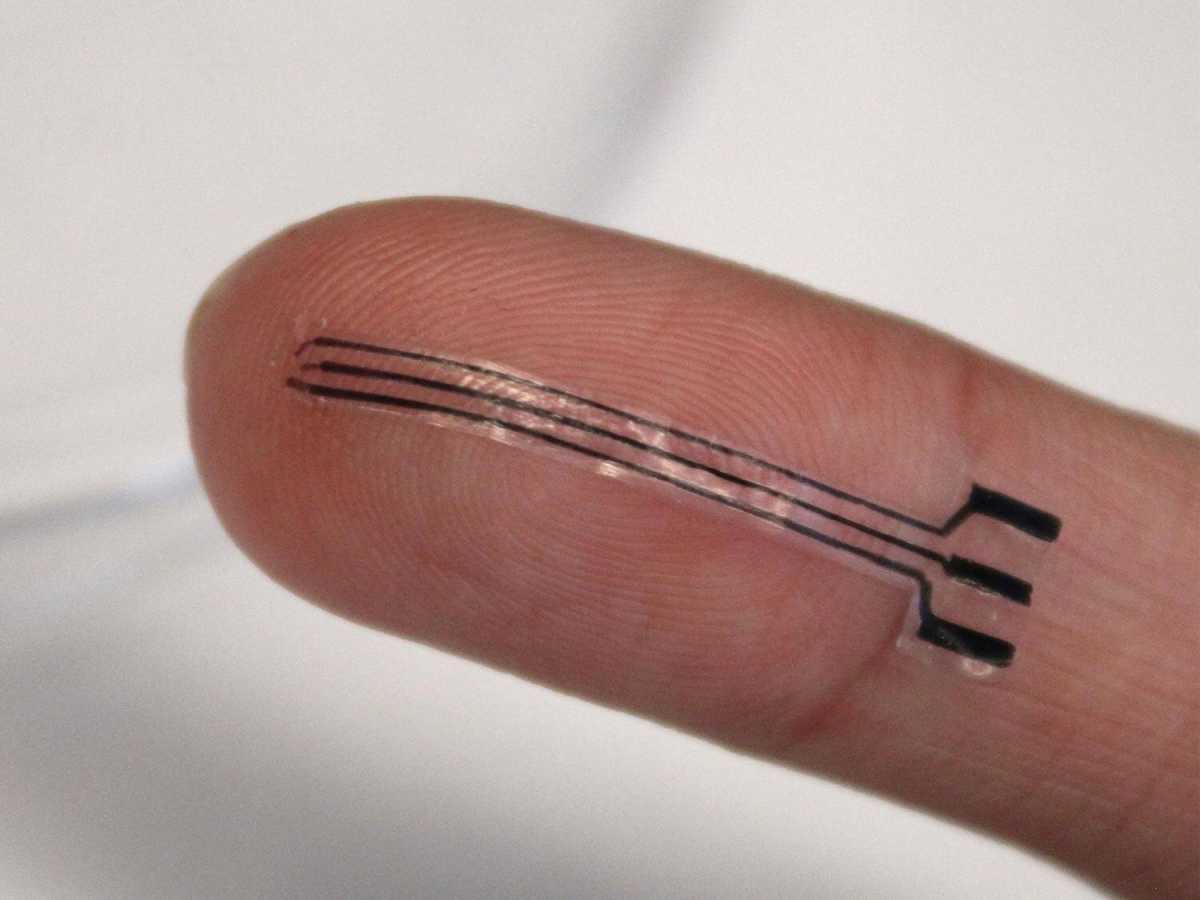
Researchers at Pennsylvania State University have developed a new class of stretchy implants designed to treat high blood pressure. These tiny devices, which can be as small as the size of a fingertip, are made from soft, stretchable materials and an adhesive component that allows them to stick painlessly to arterial tissue. According to Tao Zhou, Wormley Family Early Career Assistant Professor of Engineering Science and Mechanics at Penn State, these implants could offer a promising alternative for patients with drug-resistant hypertension.
High blood pressure, also known as hypertension, is a leading cause of heart disease in the United States, affecting nearly half of all adults. Approximately one in ten individuals with high blood pressure experience drug-resistant hypertension that can be challenging to manage. Traditional treatments often involve multiple medications and lifestyle changes, but these methods may not always provide relief.
Zhou's team has created CaroFlex, a new type of bioelectronic device specifically designed for treating hypertension. Unlike rigid metal or plastic devices used in conventional treatments, CaroFlex is made from hydrogel - a soft, jelly-like material that better integrates with the body’s tissues. The conductive hydrogels within the device transmit electrical signals to stimulate baroreceptors located in the carotid sinus, a critical area of the arterial system.
The team's research demonstrates that CaroFlex can be stretched over twice its original size without breaking and maintains strong adhesion even after extended storage. Laboratory tests showed that CaroFlex performed better than traditional electrodes in terms of electrical conductivity and compatibility with biological tissues.
To evaluate the effectiveness of CaroFlex, the researchers implanted it into the carotid sinus of rat models and monitored their blood pressure responses over a 10-minute period. Four out of five tested frequencies successfully reduced active blood pressure by an average of more than 15%. Additionally, after two weeks, there was no evidence of tissue damage or immune response around the implant.
While these results are promising, further refinement is needed before CaroFlex can be considered for human clinical trials. The next step will involve fine-tuning the device's performance and scaling up its production to prepare for potential FDA approval.
This innovative approach not only offers a non-invasive treatment option but also addresses some of the limitations associated with rigid electrodes used in current hypertension therapies. As Zhou notes, "Our lab is actively leading several developments in 3D-printed bioelectronics for use across the body," indicating that this technology has broader applications beyond treating high blood pressure.
As research continues to advance, these stretchy implants may provide a new hope for patients suffering from drug-resistant hypertension and other cardiovascular conditions.